Ethylene sulfate is an important chemical compound widely used in industrial and chemical processes. It belongs to the class of organosulfur compounds and is known for its reactive nature and usefulness in synthesis. Due to its unique structure, ethylene sulfate plays a significant role in various applications, particularly in laboratories and industrial manufacturing. Understanding its properties, uses, and safety measures is essential for professionals working with this compound.
Chemical Structure and Composition
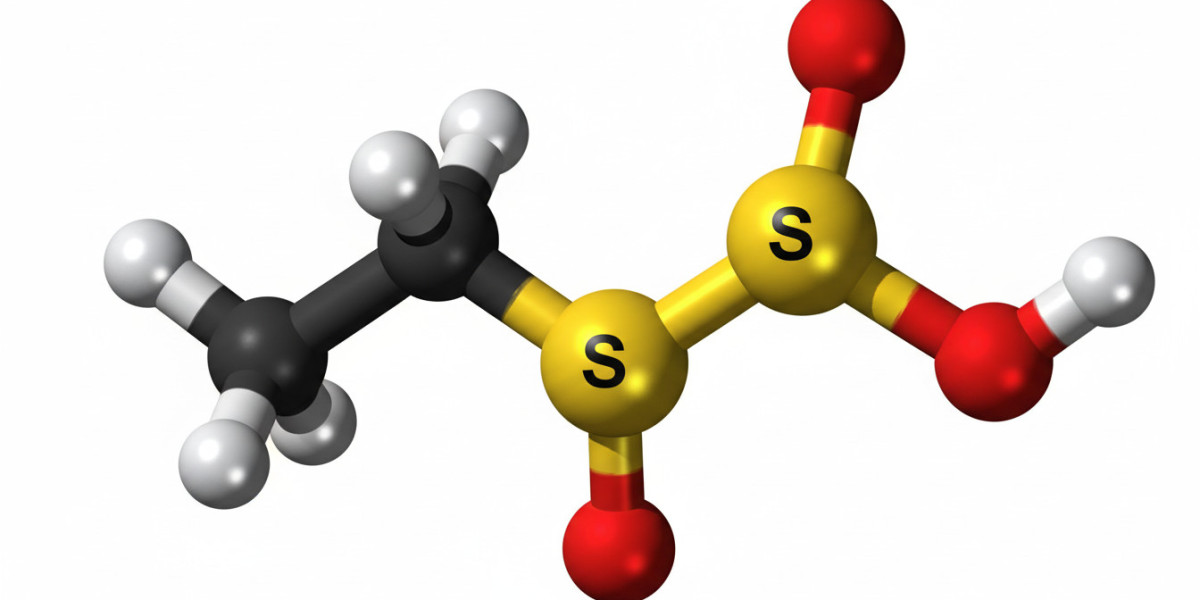
Ethylene sulfate is a cyclic sulfate ester derived from ethylene glycol. Its molecular structure consists of a three-membered ring containing sulfur and oxygen atoms, making it highly reactive. This ring strain contributes to its chemical activity, allowing it to participate in various reactions. The molecular formula of ethylene sulfate reflects its composition of carbon, hydrogen, oxygen, and sulfur atoms arranged in a compact structure.
Physical and Chemical Properties
Ethylene sulfate exhibits distinct physical and chemical properties that make it valuable in industrial applications. It is typically a colorless to pale yellow liquid with a relatively high boiling point. The compound is soluble in water and organic solvents, which enhances its versatility. Chemically, it is highly reactive due to the strained ring structure, making it prone to hydrolysis and other reactions under certain conditions.
Methods of Production
The production of ethylene sulfate usually involves the reaction of ethylene glycol with sulfur trioxide or related sulfur-containing reagents. This process forms the cyclic sulfate ester through controlled conditions. Industrial production requires careful handling due to the reactive nature of the chemicals involved. The synthesis process is designed to ensure purity and stability of the final product for its intended applications.
Industrial Applications of Ethylene Sulfate
Ethylene sulfate is widely used in the chemical industry for various purposes. It serves as an intermediate in organic synthesis, helping to produce other valuable compounds. The compound is also used in the preparation of polymers, pharmaceuticals, and specialty chemicals. Its ability to act as a reactive intermediate makes it an essential component in many manufacturing processes.
Role in Organic Chemistry
In organic chemistry, ethylene sulfate is used as a reagent for introducing sulfate groups into molecules. It participates in nucleophilic substitution reactions, where its ring structure opens to form new compounds. This property makes it useful in laboratory research and chemical synthesis. Chemists rely on ethylene sulfate for its efficiency in forming complex molecular structures.
Safety Considerations and Handling
Handling ethylene sulfate requires strict safety precautions due to its reactive and potentially hazardous nature. It can cause irritation to the skin, eyes, and respiratory system if not handled properly. Protective equipment such as gloves, goggles, and lab coats should always be used when working with this compound. Proper ventilation and storage conditions are also essential to minimize risks.
Health Effects and Toxicity
Exposure to ethylene sulfate can have adverse health effects. Inhalation or direct contact may lead to irritation and discomfort. Prolonged exposure could result in more serious health issues, depending on the concentration and duration. It is important to follow safety guidelines and limit exposure to ensure the well-being of individuals working with this chemical.
Environmental Impact
Ethylene sulfate can have an impact on the environment if not managed properly. Improper disposal may lead to contamination of water and soil. The compound can undergo chemical reactions in the environment, potentially affecting ecosystems. Therefore, industries must follow environmental regulations and guidelines for safe disposal and handling of chemical waste.
Storage and Stability
Proper storage of ethylene sulfate is crucial to maintain its stability and prevent accidents. It should be kept in tightly sealed containers, away from moisture and heat. The compound may react with water or other substances, so controlled storage conditions are necessary. Following recommended storage practices helps ensure safety and longevity of the chemical.
Advantages in Industrial Use
Ethylene sulfate offers several advantages in industrial applications. Its high reactivity allows for efficient chemical transformations, reducing processing time. The compound’s versatility makes it suitable for a wide range of uses, from synthesis to manufacturing. These benefits contribute to its popularity in the chemical industry.
Limitations and Challenges
Despite its advantages, ethylene sulfate also presents certain challenges. Its reactive nature can make handling difficult, requiring specialized equipment and training. Safety concerns and environmental considerations add to the complexity of its use. Industries must balance these factors to utilize the compound effectively while minimizing risks.
Regulatory Guidelines
Regulatory bodies have established guidelines for the safe use and handling of ethylene sulfate. These regulations aim to protect workers, consumers, and the environment. Compliance with these standards is essential for industries dealing with chemical substances. Proper documentation, labeling, and safety procedures are part of these guidelines.
Future Prospects and Research
Research on ethylene sulfate continues to explore new applications and safer handling methods. Scientists are working on developing more efficient synthesis techniques and reducing environmental impact. Advances in technology may lead to improved uses of this compound in various fields. The future of ethylene sulfate looks promising with ongoing innovation.
Conclusion
Ethylene sulfate is a valuable chemical compound with significant industrial and scientific importance. Its unique properties and reactivity make it a key component in many applications. However, proper handling, safety measures, and environmental considerations are essential to ensure its responsible use. By understanding its characteristics and applications, industries and researchers can make the most of this versatile compound.